Selank (10 mg Vial) Dosage Protocol
Quickstart Highlights
Selank is a synthetic heptapeptide analog of tuftsin with demonstrated anxiolytic and anti-asthenic (energy-boosting) properties in human clinical trials[1][2]. In Russian studies, intranasal Selank produced anxiolytic effects comparable to benzodiazepines without sedation or dependence potential[1]. This educational protocol presents a once‑daily subcutaneous approach using practical dilution for accurate insulin‑syringe measurements.
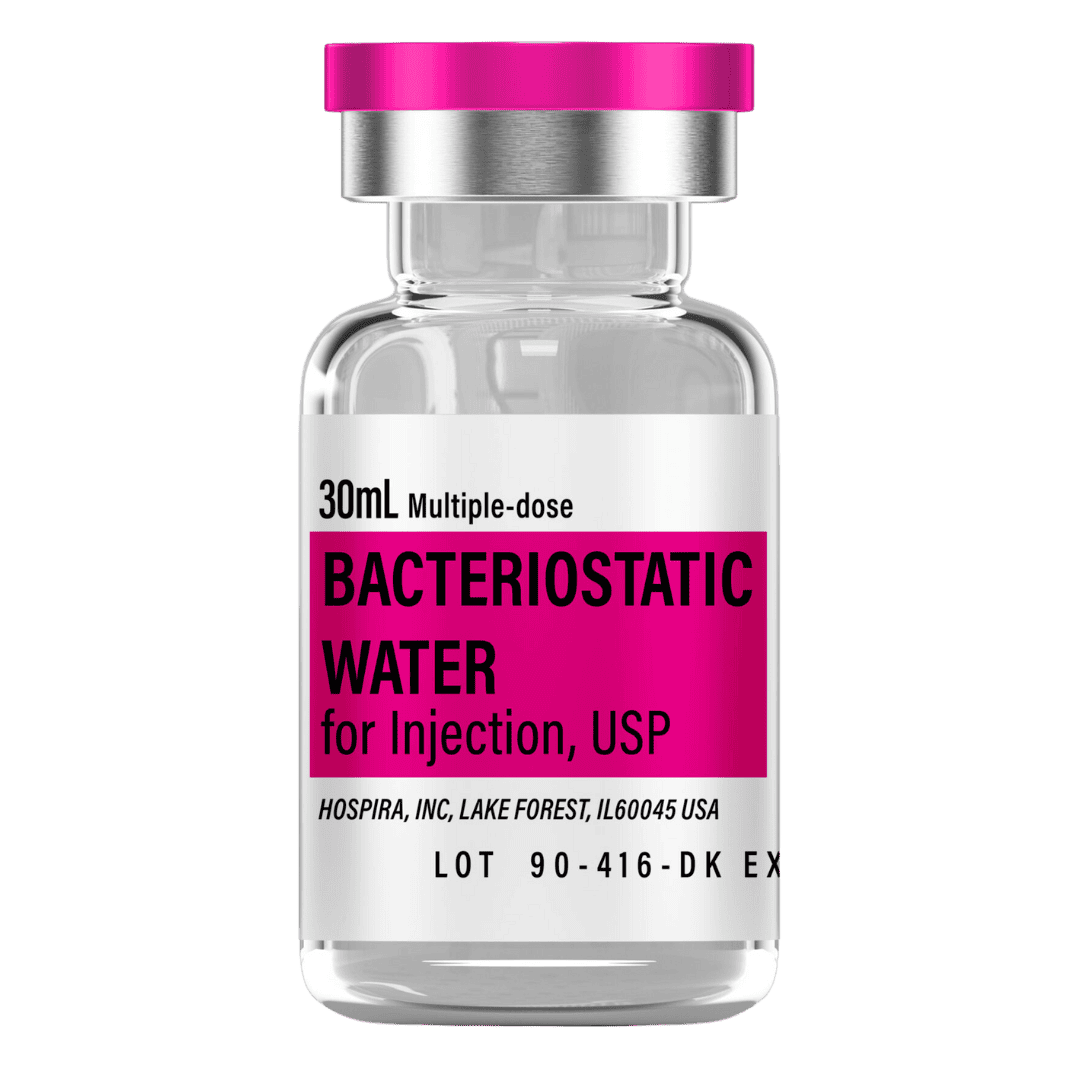
- Reconstitute: Add 3.0 mL bacteriostatic water (max vial capacity) → ~3.33 mg/mL concentration.
- Typical daily range: 300–500 mcg once daily subcutaneously (gradual titration).
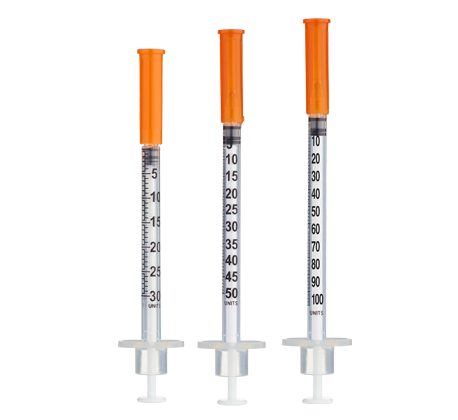
- Easy measuring: At 3.33 mg/mL, 1 unit = 0.01 mL ≈ 33.3 mcg on a U‑100 insulin syringe.
- Storage: Lyophilized: freeze at −20 °C (−4 °F); after reconstitution, refrigerate at 2–8 °C (35.6–46.4 °F); avoid freeze–thaw cycles.

Dosing & Reconstitution Guide
Educational guide for reconstitution and daily dosing
Standard / Gradual Approach (3 mL = ~3.33 mg/mL)
| Week | Daily Dose (mcg) | Units (per injection) (mL) |
|---|---|---|
| Weeks 1–2 | 300 mcg (0.3 mg) | 9 units (0.09 mL) |
| Weeks 3–4 | 500 mcg (0.5 mg) | 15 units (0.15 mL) |
Frequency: Inject once daily subcutaneously[3]. A common cycling pattern is 4 weeks on, 4 weeks off to prevent tachyphylaxis, though continuous daily use has been studied in clinical trials for up to 14 days with good tolerability[2]. Some practitioners recommend administering 5 days per week with 2-day breaks[3].
For ≤10‑unit (≤0.10 mL) administrations, consider 30‑ or 50‑unit insulin syringes for improved readability.
Reconstitution Steps
- Draw 3.0 mL bacteriostatic water with a sterile syringe.
- Inject slowly down the vial wall; avoid foaming.
- Gently swirl/roll until dissolved (do not shake vigorously as peptides are delicate).
- Label and refrigerate at 2–8 °C (35.6–46.4 °F), protected from light.
Supplies Needed
Plan based on an 8–16 week protocol with 4-week cycles and gradual titration.
-
Peptide Vials (Selank, 10 mg each):
- 8 weeks (one 4-week cycle) ≈ 2 vials
- 12 weeks (1.5 cycles) ≈ 4 vials
- 16 weeks (two complete cycles) ≈ 5 vials
-
Insulin Syringes (U‑100):
- Per week: 7 syringes (1/day)
- 8 weeks: 56 syringes
- 12 weeks: 84 syringes
- 16 weeks: 112 syringes
-
Bacteriostatic Water (10 mL bottles): Use ~3.0 mL per vial for reconstitution.
- 8 weeks (2 vials): 6 mL → 1 × 10 mL bottle
- 12 weeks (4 vials): 12 mL → 2 × 10 mL bottles
- 16 weeks (5 vials): 15 mL → 2 × 10 mL bottles
-
Alcohol Swabs: One for the vial stopper + one for the injection site each day.
- Per week: 14 swabs (2/day)
- 8 weeks: 112 swabs → recommend 2 × 100‑count boxes
- 12 weeks: 168 swabs → recommend 2 × 100‑count boxes
- 16 weeks: 224 swabs → recommend 3 × 100‑count boxes
Protocol Overview
Concise summary of the once‑daily anxiolytic/nootropic regimen.
- Goal: Support reduction of anxiety and neurasthenia symptoms while providing anti-asthenic (energy-boosting) benefits[1][2].
- Schedule: Daily subcutaneous injections for 4-week cycles with 4-week breaks (8–16 weeks total protocol).
- Dose Range: 300–500 mcg daily with gradual titration.
- Reconstitution: 3.0 mL per 10 mg vial (~3.33 mg/mL) for accurate unit measurements.
- Storage: Lyophilized frozen; reconstituted refrigerated; avoid repeated freeze–thaw.
Dosing Protocol
Suggested daily titration approach.
- Start: 300 mcg daily for weeks 1–2; clinicians treating cognitive or anxiety issues often begin at this level[3].
- Target: 500 mcg daily by weeks 3–4 if lower doses are well-tolerated.
- Frequency: Once per day (subcutaneous); some protocols use 5 days per week with 2-day breaks[3].
- Cycle Pattern: 4 weeks on, 4 weeks off to prevent tachyphylaxis; repeat as needed.
- Timing: Any consistent time; rotate injection sites to prevent irritation.
Storage Instructions
Proper storage preserves peptide quality.
- Lyophilized: Store at −20 °C (−4 °F) for long-term stability (several months); refrigeration at 2–8 °C (35.6–46.4 °F) acceptable for shorter periods[10].
- Reconstituted: Refrigerate at 2–8 °C (35.6–46.4 °F); stable for approximately 1 month with minimal potency loss[10]. Prepare aliquots if needed and avoid freeze–thaw cycles.
- Allow vials to reach room temperature before opening to reduce condensation and moisture uptake.
Important Notes
Practical considerations for consistency and safety.
- Use new sterile insulin syringes for each injection; dispose in a sharps container immediately after use[9].
- Rotate injection sites (abdomen, thighs, upper arms) systematically to reduce local irritation and prevent tissue changes[7].
- Inject slowly and steadily; wait a few seconds before withdrawing the needle.
- Document daily dose, timing, and site rotation to maintain consistency throughout the protocol.
- Multi-use bacteriostatic water vials should be dated upon opening and discarded within 28 days[9].
How This Works
Selank is a synthetic analog of the immunomodulatory peptide tuftsin, consisting of the sequence Thr-Lys-Pro-Arg-Pro-Gly-Pro. In Russian clinical trials for anxiety disorders and neurasthenia, intranasal Selank produced anxiolytic effects comparable to benzodiazepines while also providing anti-asthenic (energy-boosting) benefits without sedative side effects or dependence potential[1][2]. Preclinical studies in primates demonstrated that intranasal Selank eliminated fear and anxiety behaviors with long-term stabilizing effects on neurobehavior[4]. Rodent studies confirmed robust anxiolytic efficacy comparable to classical tranquilizers[5]. While most published human data utilized intranasal dosing (the original formulation), subcutaneous administration has shown effectiveness in research settings with consistent absorption and ease of dose measurement[3]. A 14-day continuous course was well-tolerated and effective in human trials[2], and even high-dose applications have been explored for conditions such as attenuating opioid withdrawal symptoms in animal models[5].
Potential Benefits & Side Effects
Observations from clinical and preclinical literature.
- Produces anxiolytic effects comparable to benzodiazepines in human clinical trials without sedation or dependence risk[1].
- Provides anti-asthenic (energy-boosting) benefits alongside anxiety reduction[2].
- Induces lasting neurobehavioral changes; primate studies showed elimination of fear/anxiety behaviors with long-term stabilizing effects[4].
- Generally well tolerated in clinical studies; a 14-day continuous course was well-tolerated and effective in human trials[2].
- Occasional mild injection-site reactions (redness, minor irritation) may occur with subcutaneous administration; proper site rotation minimizes this risk.
- Lacks sedative side effects typical of classical anxiolytics, allowing for daytime use without impairment[1].
- Has been explored at higher doses for experimental applications such as attenuating opioid withdrawal symptoms, indicating a wide therapeutic range[5].
Lifestyle Factors
Complementary strategies for optimal cognitive and anxiolytic outcomes.
- Maintain consistent sleep hygiene and aim for 7–9 hours of quality sleep to support neurological function and stress resilience.
- Incorporate regular physical activity; both aerobic exercise and resistance training support mood regulation and cognitive performance.
- Practice stress-management techniques such as mindfulness, meditation, or breathwork to complement Selank’s anxiolytic effects.
- Support nutritional status with adequate protein, omega-3 fatty acids, and micronutrients essential for neurotransmitter synthesis.
- Minimize alcohol and stimulant use, which can interfere with anxiety management and neurological stability.
Injection Technique
General subcutaneous guidance from clinical best-practice resources[7][8].
- Wash hands thoroughly and prepare a clean work surface.
- Clean the vial stopper with an alcohol swab; allow to dry completely.
- Draw the appropriate volume into a sterile insulin syringe; remove air bubbles by flicking the syringe and expelling air.
- Clean the injection site with a fresh alcohol swab; allow skin to dry to prevent stinging.
- Pinch a skinfold of subcutaneous tissue; insert the needle at 90° into the subcutaneous layer[7].
- Do not aspirate for subcutaneous injections; inject slowly and steadily[8].
- Withdraw the needle straight out after injection; apply gentle pressure if minor bleeding occurs.
- Dispose of the used syringe immediately in an appropriate sharps container; never reuse needles or syringes[9].
- Rotate sites systematically (abdomen at least 2 inches from navel, front/outer thighs, upper arms) to avoid lipohypertrophy[7].
Recommended Source
We recommend Pure Lab Peptides for high‑purity Selank (10 mg).
Why Pure Lab Peptides?
- High‑purity, third‑party‑tested lots with batch Certificates of Analysis (COAs).
- Consistent, ISO‑aligned handling and comprehensive documentation.
- Reliable fulfillment with proper cold‑chain management to maintain peptide integrity.
- Transparent sourcing and quality control procedures for research-grade peptides.
Important Note
This content is intended for therapeutic educational purposes only and does not constitute medical advice, diagnosis, or treatment. Selank is for research use only and not intended for human consumption. Always consult qualified healthcare professionals before considering any peptide therapy.
References
-
PubMed
— Clinical trial of Selank for generalized anxiety disorder (intranasal, human study) -
PubMed
— Selank for neurasthenia: 14-day clinical trial demonstrating anxiolytic and anti-asthenic effects (human study) -
Hindawi BioMed Research
— Intranasal Selank administration review: neurobehavioral effects and clinical applications (review article) -
Springer
— Primate studies: intranasal Selank eliminates anxiety behaviors with long-term effects (preclinical) -
Springer
— Selank attenuates opioid withdrawal symptoms in animal models (preclinical dosing study) -
CDC
— Vaccine administration best practices: subcutaneous injection technique and site selection -
Cleveland Clinic
— Subcutaneous injection guide: technique, sites, and rotation principles -
NCBI Bookshelf
— Clinical procedures: injection preparation, aseptic technique, and administration standards -
CDC
— Injection safety guidelines: single-use devices, sharps disposal, and preventing unsafe practices -
NIBSC
— Peptide storage guidelines: lyophilized and reconstituted stability considerations -
PMC
— Pharmacologic review: subcutaneous drug delivery and absorption considerations -
Pure Lab Peptides
— Selank (10 mg) product page: quality documentation and batch COAs