Mazdutide (10 mg Vial) Dosage Protocol
Quickstart Highlights
Mazdutide (IBI362) is a long‑acting dual GLP‑1/glucagon receptor agonist developed for chronic weight management and type 2 diabetes.[1][2] Phase 2 and 3 clinical trials have demonstrated substantial weight loss (6–14% reduction over 24–48 weeks) with mostly mild gastrointestinal side effects.[2][3][5] This educational protocol presents a once‑weekly subcutaneous approach with practical dilution for clear insulin‑syringe measurements.
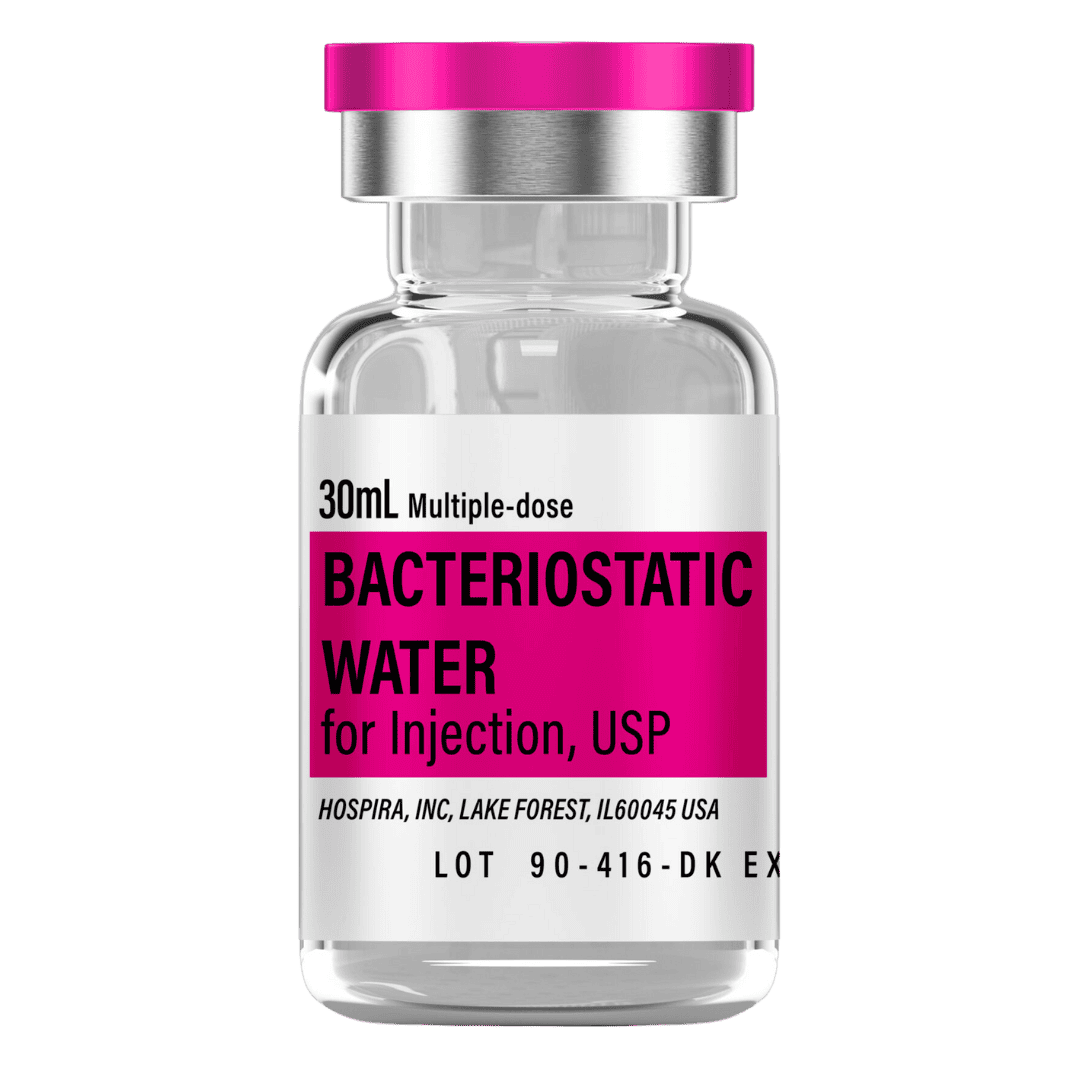
- Reconstitute: Add 3.0 mL bacteriostatic water → ~3.33 mg/mL concentration (standard protocol).
- Typical weekly range: 2.5–5 mg once weekly with gradual titration.
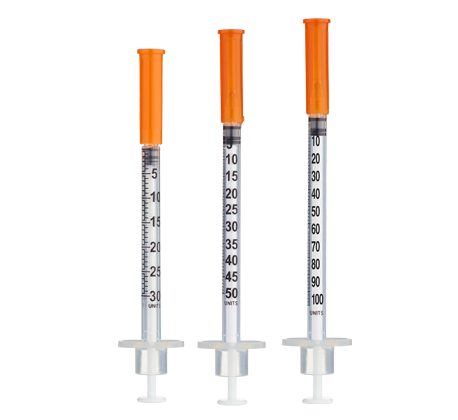
- Easy measuring: At 3.33 mg/mL, 1 unit = 0.01 mL ≈ 33.3 mcg on a U‑100 insulin syringe.
- Storage: Lyophilized: freeze at −20 °C (−4 °F); after reconstitution, refrigerate at 2–8 °C (35.6–46.4 °F); avoid freeze–thaw cycles.[7]

Dosing & Reconstitution Guide
Educational guide for reconstitution and weekly dosing
Standard / Gradual Titration (3.0 mL = ~3.33 mg/mL)
| Week | Weekly Dose | Units (per injection) (mL) |
|---|---|---|
| Weeks 1–4 | 2.5 mg (2,500 mcg) | 75 units (0.75 mL) |
| Weeks 5–8+ | 5 mg (5,000 mcg) | 150 units (1.50 mL) |
Frequency: Inject once weekly subcutaneously. This schedule mirrors clinical trial protocols with gradual titration to improve tolerability.[1][2] The 3.0 mL dilution provides practical unit measurements for standard maintenance doses.
Reconstitution Steps
- Draw 3.0 mL bacteriostatic water with a sterile syringe.
- Inject slowly down the vial wall; avoid foaming.
- Gently swirl until dissolved (do not shake vigorously).
- Label with date and refrigerate at 2–8 °C (35.6–46.4 °F), protected from light.
Advanced / High‑Dose Protocol (2.0 mL = 5.0 mg/mL)
| Week | Weekly Dose | Units (per injection) (mL) | Notes |
|---|---|---|---|
| Weeks 1–4 | 5 mg (5,000 mcg) | 100 units (1.00 mL) | 0.5 vials |
| Weeks 5–8 | 7.5 mg (7,500 mcg) | 150 units (1.50 mL) | 0.75 vials |
| Weeks 9–12+ | 10 mg (10,000 mcg) | 200 units (2.00 mL) | 1 vial |
Caution: High‑dose protocols (7.5–10 mg weekly) have been explored in phase 1b research but require careful monitoring and clinical oversight.[1] Doses above 6 mg increase the risk of gastrointestinal side effects. The 2.0 mL reconstitution provides higher concentration to reduce injection volume. For doses requiring >3.0 mL total volume, split into two separate injections at different sites.
Supplies Needed
Plan based on an 8–16 week weekly protocol with gradual titration to 5 mg maintenance.
-
Peptide Vials (Mazdutide, 10 mg each):
- 8 weeks ≈ 3 vials (Weeks 1–4: 10 mg total = 1 vial; Weeks 5–8: 20 mg total = 2 vials)
- 12 weeks ≈ 5 vials (Weeks 1–4: 10 mg = 1 vial; Weeks 5–12: 40 mg = 4 vials)
- 16 weeks ≈ 7 vials (Weeks 1–4: 10 mg = 1 vial; Weeks 5–16: 60 mg = 6 vials)
-
Insulin Syringes (U‑100, 1 mL or 3 mL capacity):
- Per week: 1 syringe (once‑weekly dosing)
- 8 weeks: 8 syringes
- 12 weeks: 12 syringes
- 16 weeks: 16 syringes
-
Bacteriostatic Water (10 mL bottles): Use 3.0 mL per vial for standard reconstitution.
- 8 weeks (3 vials): 9 mL → 1 × 10 mL bottle
- 12 weeks (5 vials): 15 mL → 2 × 10 mL bottles
- 16 weeks (7 vials): 21 mL → 3 × 10 mL bottles
-
Alcohol Swabs: One for the vial stopper + one for the injection site each week.
- Per week: 2 swabs
- 8 weeks: 16 swabs → recommend 1 × 100‑count box
- 12 weeks: 24 swabs → recommend 1 × 100‑count box
- 16 weeks: 32 swabs → recommend 1 × 100‑count box
Protocol Overview
Concise summary of the once‑weekly regimen.
- Goal: Support chronic weight management and metabolic improvement through dual GLP‑1/glucagon receptor activation.[2][3]
- Schedule: Once‑weekly subcutaneous injections for 8–12 weeks minimum (clinical trials ranged from 12–48 weeks).
- Dose Range: 2.5–5 mg weekly with gradual titration; advanced protocols may reach 10 mg.
- Reconstitution: 3.0 mL per 10 mg vial (~3.33 mg/mL) for standard dosing; 2.0 mL for higher concentrations if needed.
- Storage: Lyophilized frozen at −20 °C (−4 °F); reconstituted refrigerated at 2–8 °C (35.6–46.4 °F); discard after 28 days.[7][9]
Dosing Protocol
Suggested weekly titration approach.
- Start: 2.5 mg once weekly for first 4 weeks to establish tolerability.
- Increase: Advance to 5 mg weekly at week 5; maintain for remainder of protocol (weeks 5–12+).
- Frequency: Once per week (subcutaneous injection).
- Cycle Length: Minimum 8 weeks; clinical evidence supports 12–48 week protocols.
- Timing: Administer on the same day each week; rotate injection sites to avoid irritation.
Storage Instructions
Proper storage preserves peptide stability and potency.
- Lyophilized: Store at −20 °C (−4 °F) or below in dry, dark conditions; protect from moisture.
- Reconstituted: Refrigerate at 2–8 °C (35.6–46.4 °F); use bacteriostatic water for multi‑dose stability.[9]
- Important: Do not freeze reconstituted solution; avoid freeze–thaw cycles which can denature the peptide.[7]
- Discard: Dispose of reconstituted vials after 28 days or if cloudiness/particulates appear.
Important Notes
Practical considerations for safe and effective administration.
- Use a new sterile insulin syringe for each weekly injection; dispose in a proper sharps container.[9]
- Rotate injection sites weekly (abdomen, thighs, upper arms) to minimize local irritation and lipoatrophy.[8][10]
- For doses requiring >3 mL volume, split into two separate injections at different sites for comfort and absorption.
- Allow reconstituted vials to reach room temperature before withdrawing dose to reduce viscosity.
- Document dose, injection site, and any side effects to track response and optimize protocol.
How This Works
Mazdutide functions as a dual agonist at both GLP‑1 and glucagon receptors, combining the appetite‑suppressing and glucose‑lowering effects of GLP‑1 with the energy‑expenditure benefits of glucagon activation.[2][6] This dual mechanism distinguishes it from single‑target GLP‑1 agonists and has shown robust weight loss efficacy in clinical trials. Phase 2 studies demonstrated 6.7–11.3% body weight reduction at 3–6 mg weekly doses over 24 weeks.[2] A 48‑week phase 3 trial reported sustained weight loss of 11–14% with 4–6 mg weekly maintenance.[3] In type 2 diabetes populations, mazdutide achieved meaningful HbA1c reductions (~1.7%) alongside ~7% weight loss.[4] The extended half‑life enables convenient once‑weekly dosing with steady pharmacologic exposure.
Potential Benefits & Side Effects
Observations from clinical trial evidence.
Potential Benefits
- Significant body weight reduction (6–14% over 24–48 weeks) in obesity trials.[2][3][5]
- Meaningful HbA1c improvement (~1.7% reduction) in type 2 diabetes populations.[4]
- Favorable metabolic effects including improved lipid profiles and glucose control.
- Convenient once‑weekly dosing improves adherence compared to daily regimens.
Common Side Effects
- Gastrointestinal effects (nausea, vomiting, diarrhea, constipation) are the most common adverse events, typically mild‑to‑moderate and diminishing with continued use.[5]
- Injection‑site reactions (redness, swelling, mild discomfort) may occur; rotate sites to minimize.
- Gradual dose titration significantly improves tolerability and reduces discontinuation rates.[1][2]
Lifestyle Factors
Complementary strategies for optimal outcomes.
- Maintain a balanced, nutrient‑dense diet with adequate protein to support lean mass during weight loss.
- Combine regular resistance training with aerobic exercise to preserve muscle and enhance metabolic adaptations.
- Prioritize consistent sleep (7–9 hours nightly) and stress management to support adherence and hormonal balance.
- Stay well‑hydrated and monitor electrolyte intake, especially during initial titration when GI effects may be present.
Injection Technique
Subcutaneous injection guidance based on clinical best practices.[8][10]
- Clean the vial stopper and injection site with alcohol swabs; allow to air‑dry completely.
- Use aseptic technique: wipe stoppers, use sterile needles, and avoid touching needle tip.
- Pinch a skinfold and insert the needle at 45–90° into subcutaneous fatty tissue (not muscle).
- Do not aspirate for subcutaneous injections; inject slowly and steadily over 5–10 seconds.
- Withdraw needle and apply gentle pressure with a clean swab; do not rub vigorously.
- Rotate injection sites systematically: abdomen (at least 2 inches from navel), front/outer thighs, or outer upper arms.
- For volumes >3 mL, divide dose into two injections at separate sites for comfort and absorption.
Recommended Source
We recommend Pure Lab Peptides for high‑purity Mazdutide (10 mg).
Why Pure Lab Peptides?
- High‑purity, third‑party‑tested peptides with batch‑specific Certificates of Analysis (COAs).
- Consistent quality control and ISO‑aligned handling procedures.
- Reliable cold‑chain shipping and proper storage documentation.
- Dedicated customer support for research peptide applications.
Important Note
This content is intended for therapeutic educational purposes only and does not constitute medical advice, diagnosis, or treatment. Mazdutide is an investigational peptide not approved for prescription use in most countries (as of 2025, it has received initial approval in China for obesity treatment). All information is provided for research and educational purposes only. Consult qualified healthcare professionals before considering any peptide protocol.
References
-
EClinicalMedicine (PubMed)
— Phase 1b trial: Safety and efficacy of mazdutide 9–10 mg in Chinese adults with overweight/obesity -
Nature Communications
— Phase 2 randomized controlled trial of mazdutide in Chinese overweight/obese adults -
New England Journal of Medicine
— Phase 3 trial: Once‑weekly mazdutide in Chinese adults with obesity or overweight (48‑week results) -
Diabetes Care (PubMed)
— Phase 2 trial: Efficacy and safety of mazdutide in Chinese patients with type 2 diabetes (20‑week results) -
Frontiers in Endocrinology
— Systematic review and meta‑analysis: Efficacy and safety of mazdutide on weight loss (diabetic and non‑diabetic patients) -
International Journal of Obesity (Nature)
— Review: Pipeline for future obesity medications (including mazdutide dual agonist mechanism) -
Bachem Peptide Technical Guide
— Handling and storage guidelines for peptides (lyophilized and in solution) -
CDC
— Vaccine administration: Subcutaneous injection technique (angle, site selection, no aspiration) -
CDC
— Injection safety for healthcare personnel (multi‑dose vial handling, aseptic technique) -
NCBI Bookshelf
— Clinical procedures: Subcutaneous injections (best practices and site rotation) -
Pure Lab Peptides
— Mazdutide (10 mg) product page (quality documentation and batch COAs)