SLU-PP-332 (5 mg Vial) Dosage Protocol
Quickstart Highlights
PRECLINICAL RESEARCH COMPOUND: SLU-PP-332 is a synthetic pan-agonist of estrogen-related receptors (ERRα/β/γ) studied exclusively in murine models for its ability to activate an aerobic-exercise gene program, enhance mitochondrial function, and improve endurance and cardiometabolic phenotypes[1][2][3]. No human trials have been conducted as of November 2025. This educational protocol reflects published murine regimens using intraperitoneal administration.
- Evidence Base: Preclinical only—in vivo mouse studies with intraperitoneal dosing (25–50 mg/kg twice daily).
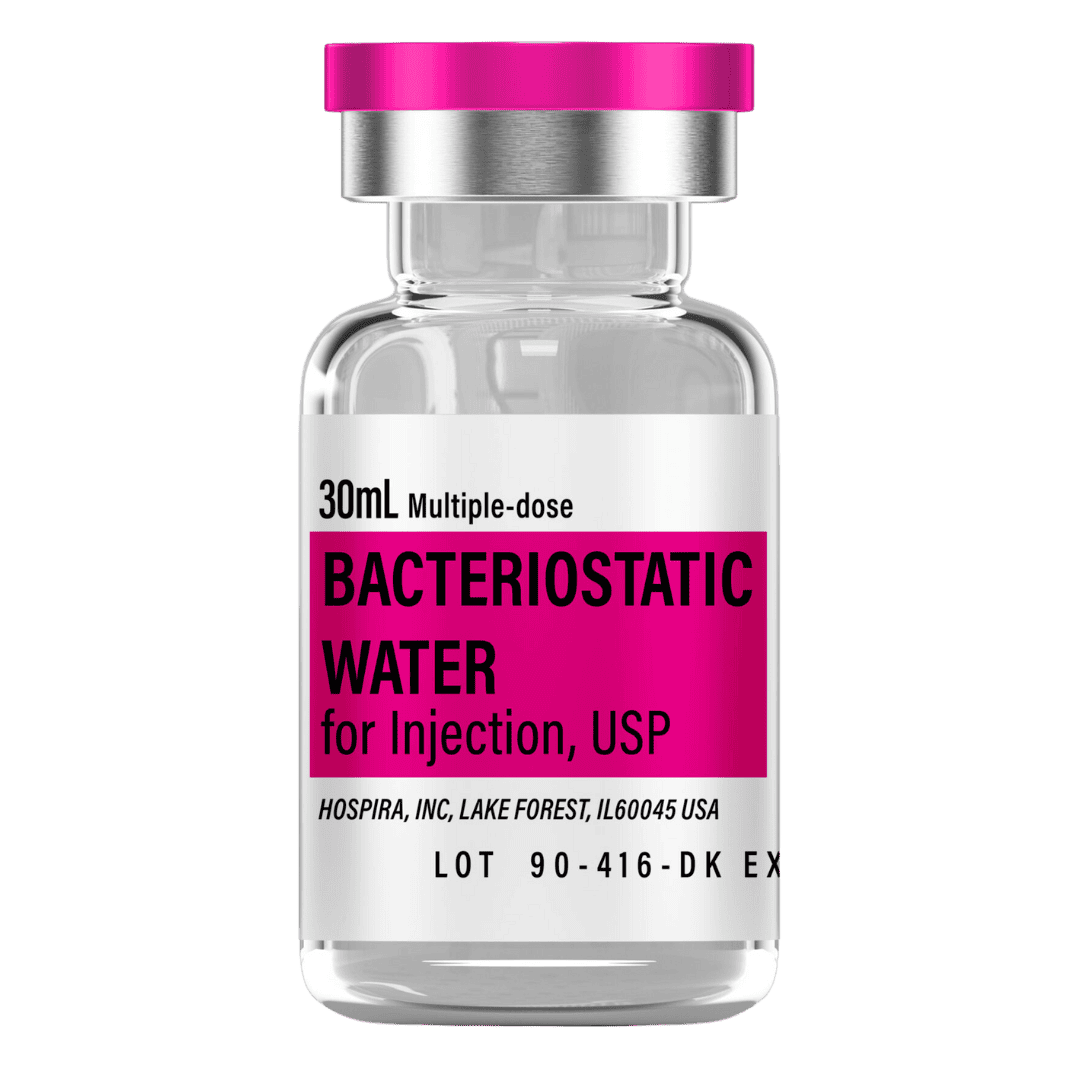
- Reconstitute: Add 3.0 mL bacteriostatic water → ~1.67 mg/mL concentration.
- Murine dose range: 1250–2500 mcg daily (for a 25 g mouse), administered in two divided doses.
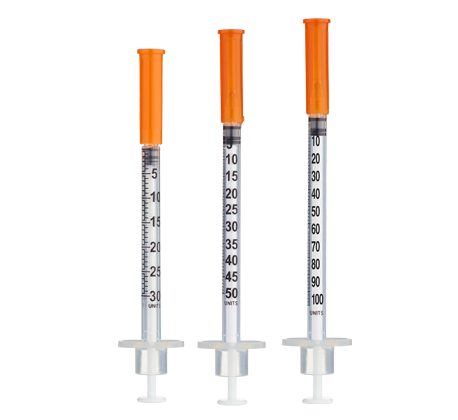
- Easy measuring: At 1.67 mg/mL, 1 unit = 0.01 mL ≈ 16.7 mcg on a U-100 insulin syringe.
- Storage: Lyophilized: −20 °C (−4 °F); reconstituted: 2–8 °C (35.6–46.4 °F).

Dosing & Reconstitution Guide
Educational guide based on published murine regimens
Murine-Equivalent Protocol (3 mL = ~1.67 mg/mL)
| Phase | Daily Dose (mcg) | Per-Injection Dose (mcg) | Units per Injection (mL) |
|---|---|---|---|
| Weeks 1–2 | 1250 mcg | 625 mcg | 37.5 units (0.375 mL) |
| Weeks 3–8 | 2500 mcg | 1250 mcg | 75 units (0.75 mL) |
Route & Frequency: Published murine studies used intraperitoneal administration twice daily (25–50 mg/kg)[1][2][3]. Daily mcg values above are expressed for a 25 g mouse. No reputable sources support once-daily subcutaneous use for this compound.
Reconstitution Steps
- Draw 3.0 mL bacteriostatic water with a sterile syringe.
- Inject slowly down the vial wall; avoid foaming.
- Gently swirl/roll until dissolved (do not shake).
- Label and refrigerate at 2–8 °C (35.6–46.4 °F), protected from light.
Supplies Needed
Based on an 8–16 week murine-equivalent protocol with twice-daily administration.
-
Peptide Vials (SLU-PP-332, 5 mg each):
- 8 weeks ≈ 25 vials
- 12 weeks ≈ 39 vials
- 16 weeks ≈ 53 vials
-
Insulin Syringes (U-100):
- Per week: 14 syringes (2/day)
- 8 weeks: 112 syringes
- 12 weeks: 168 syringes
- 16 weeks: 224 syringes
-
Bacteriostatic Water (10 mL bottles): Use ~3.0 mL per vial for reconstitution.
- 8 weeks (25 vials): 75 mL → 8 × 10 mL bottles
- 12 weeks (39 vials): 117 mL → 12 × 10 mL bottles
- 16 weeks (53 vials): 159 mL → 16 × 10 mL bottles
-
Alcohol Swabs: One for the vial stopper + one for the injection site each administration.
- Per week: 28 swabs (4/day)
- 8 weeks: 224 swabs → recommend 3 × 100-count boxes
- 12 weeks: 336 swabs → recommend 4 × 100-count boxes
- 16 weeks: 448 swabs → recommend 5 × 100-count boxes
Protocol Overview
Concise summary of the murine-based regimen.
- Evidence Status: Preclinical only—no human trials exist[1][11].
- Mechanism: Pan-ERR agonist; activates aerobic-exercise gene program and enhances mitochondrial function.
- Published Route: Intraperitoneal administration in mice (25–50 mg/kg twice daily).
- Cycle Length: 8 weeks in published studies.
- Reconstitution: 3.0 mL per 5 mg vial (~1.67 mg/mL).
- Storage: Lyophilized frozen; reconstituted refrigerated.
Dosing Protocol
Murine-equivalent dosing framework.
- Initial Phase (Weeks 1–2): 1250 mcg daily (625 mcg per injection, twice daily).
- Maintenance Phase (Weeks 3–8): 2500 mcg daily (1250 mcg per injection, twice daily).
- Frequency: Twice per day, reflecting published murine regimens[1][2][3].
- Route: Intraperitoneal (as studied in preclinical models).
- Timing: Consistent intervals between administrations.
Storage Instructions
Proper storage preserves compound stability[7][8][9][10].
- Lyophilized: Store at −20 °C (−4 °F) in dry, dark conditions; minimize moisture exposure.
- Reconstituted: Refrigerate at 2–8 °C (35.6–46.4 °F); avoid freeze–thaw cycles.
- Allow vials to reach room temperature before opening to reduce condensation.
- Protect from light during storage and handling.
Important Notes
Research compound considerations.
- No human data: SLU-PP-332 has not been tested in human subjects; all evidence is from murine models.
- Use new sterile syringes for each administration; dispose in a sharps container.
- Rotate injection sites to reduce local irritation.
- Document administration times, doses, and any observations.
- This compound is for research purposes only—not for human consumption.
How This Works
SLU-PP-332 is a synthetic small-molecule pan-agonist of the estrogen-related receptors (ERRα, ERRβ, and ERRγ), with strongest activity at ERRα[1][11]. In murine studies, acute administration activated an aerobic-exercise transcriptional program, increased mitochondrial respiration and fatty-acid oxidation in skeletal muscle, and improved endurance capacity and cardiometabolic phenotypes[3]. The compound acts as an “exercise mimetic” by engaging metabolic pathways typically activated during physical training, without requiring the mechanical stress of actual exercise. However, all evidence to date is preclinical—no human trials have been conducted.
Preclinical Observations
Findings from murine studies only.
- Metabolic effects: Enhanced mitochondrial function, increased fatty-acid oxidation, improved glucose tolerance in diet-induced obese mice[2].
- Cardiac benefits: Ameliorated heart failure through enhanced cardiac fatty-acid metabolism and mitochondrial function in preclinical models[3].
- Endurance enhancement: Increased treadmill running capacity and muscle oxidative capacity in mice[1].
- Renal effects: Reversed mitochondrial dysfunction and inflammation in aging kidney models[4].
- Human safety unknown: No data on human tolerability, pharmacokinetics, or adverse effects exist.
Research Considerations
Contextual factors for preclinical research.
- SLU-PP-332 was studied alongside exercise training in some murine models, suggesting potential synergistic effects.
- Diet composition (standard chow vs. high-fat diet) influenced metabolic outcomes in published studies.
- Environmental controls (temperature, housing conditions) should be standardized in research protocols.
- Compound stability and handling procedures are critical for reproducible results.
Administration Technique
General subcutaneous guidance (note: published studies used intraperitoneal route)[5][6].
- Clean the vial stopper and skin with alcohol; allow to dry completely.
- For subcutaneous administration: pinch a skinfold; insert needle at 45–90° into subcutaneous tissue.
- Do not aspirate for subcutaneous injections; inject slowly and steadily.
- Rotate sites systematically (abdomen, thighs, upper arms) to avoid lipohypertrophy.
- Note: Published murine studies used intraperitoneal administration; subcutaneous route is not validated for this compound.
Recommended Source
We recommend Pure Lab Peptides for research-grade SLU-PP-332 (5 mg).
Why Pure Lab Peptides?
- High-purity research compounds with third-party testing.
- Batch-specific certificates of analysis (COAs) available.
- ISO-aligned handling and documentation protocols.
- Reliable cold-chain fulfillment for compound integrity.
Important Note
This content is intended for therapeutic educational purposes only and does not constitute medical advice, diagnosis, or treatment. SLU-PP-332 is a preclinical research compound with no human safety or efficacy data. This information is for research and educational purposes only—not for human consumption.
References
-
ACS Chemical Biology (2023)
— Synthetic ERRα/β/γ Agonist Induces an ERRα-Dependent Acute Aerobic Exercise Response and Enhances Exercise Capacity -
Journal of Pharmacology and Experimental Therapeutics (2024)
— A Synthetic ERR Agonist Alleviates Metabolic Syndrome -
Circulation (2024)
— Novel Pan-ERR Agonists Ameliorate Heart Failure Through Enhancing Cardiac Fatty Acid Metabolism and Mitochondrial Function -
American Journal of Pathology (2023)
— Estrogen-Related Receptor Agonism Reverses Mitochondrial Dysfunction and Inflammation in the Aging Kidney -
CDC (2024)
— Vaccine Administration: Subcutaneous Injection (Job Aid) -
CDC (2024)
— Vaccine Administration—Best Practices (Subcutaneous Injections) -
Advanced Drug Delivery Reviews (2023)
— Strategies for overcoming protein and peptide instability in biodegradable drug delivery systems -
Journal of Chromatography B (2023)
— Tutorial review for peptide assays: An ounce of prevention is worth a pound of cure? -
Clinical Chemistry (2016)
— Recommendations for the generation, quantification, storage and handling of peptides used for mass spectrometry-based assays -
Journal of Pharmaceutical Sciences (2012)
— Use of glass transitions in carbohydrate excipient design for lyophilized biopharmaceuticals -
Acta Pharmacologica Sinica (2015)
— The multiple universes of estrogen-related receptor α and γ in metabolic control and related diseases -
Pure Lab Peptides
— SLU-PP-332 5mg product page (supplier and batch documentation)